📹
🧪
🌡️
♻️
🧩
🔬
💡
⚡
🔥 High-Temp Fuel Cell Testing
🧪 Electrolyte–Carbon Interfaces
📹 Python Video Processing
📊 Contact Angle Analysis
🔋 Fuel Cell Optimization
From Methane to Clean Power
This project was launched under CanmetENERGY’s efforts to repurpose natural gas through thermal cracking, turning harmful methane emissions into useful carbon and hydrogen streams. Instead of releasing these byproducts, the process captures their value through two parallel pathways. The solid carbon is directed into a Direct Carbon Fuel Cell (DCFC), where it generates electricity while producing a pure, sequesterable CO₂ stream. In parallel, the liberated hydrogen is purified and used in hydrogen fuel cells, supporting both clean power generation and the hydrogen economy.
By linking these pathways, the project demonstrates how thermal cracking can become a cornerstone of decarbonization: carbon transformed into controlled energy output, hydrogen into a versatile clean fuel, and both working together to lower emissions while improving energy efficiency.
Hydraulic press used to compact electrolyte
powders into pellets for furnace testing.
Engineering Objectives
This project was designed with three primary technical goals:
- Carbon–Electrolyte Optimization – Identify the most effective pairing of carbon feedstock and electrolyte materials to maximize electrode–electrolyte contact and reaction efficiency at operating temperatures approaching 800 °C.
- Continuous Fuel Delivery – Develop strategies to continuously feed solid carbon fuel into the DCFC, overcoming challenges in maintaining consistent carbon contact during sustained operation.
- Cell Stacking for Voltage Output – Explore methods to stack multiple single cells into a modular unit to increase voltage output, while testing material durability and interfacial stability under repeated cycling.
Within this broader scope, my focus centered on carbon–electrolyte optimization. I conducted wettability experiments to evaluate how different carbon materials interact with molten electrolytes, aiming to identify pairings that promote maximum surface contact and stable reaction kinetics. My personal learning goal was to deepen my understanding of fuel cell chemistry, particularly electrolyte behavior and its role in ionic conduction.

Simulating Fuel Cell Conditions
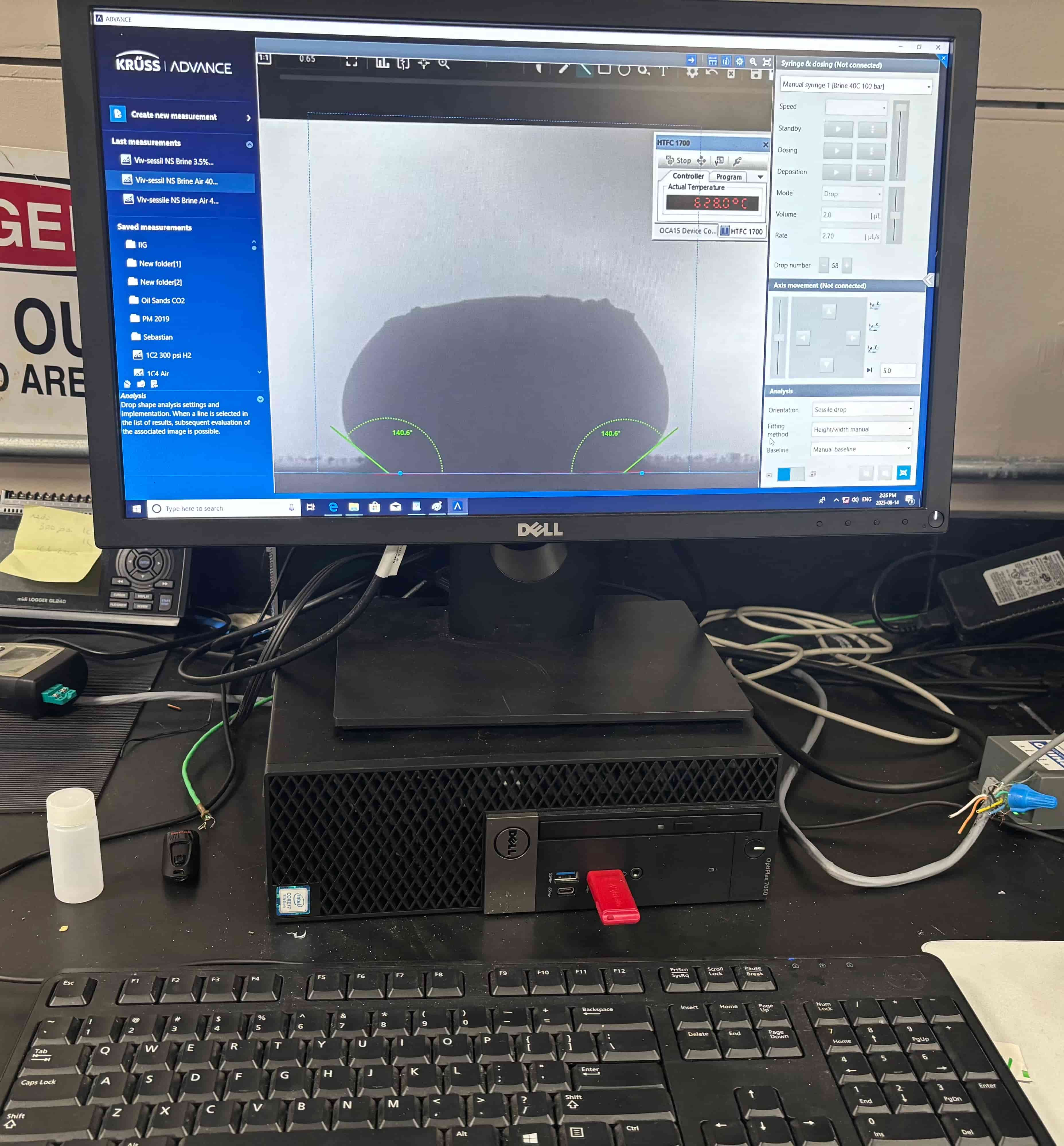
To simulate the extreme operating conditions of a Direct Carbon Fuel Cell (DCFC), we employed a specialized tensiometer system developed at Natural Resources Canada. This instrument functions as a high-temperature tube furnace, capable of reaching up to 1500 °C, making it well-suited for reproducing the molten electrolyte environment of a working DCFC.
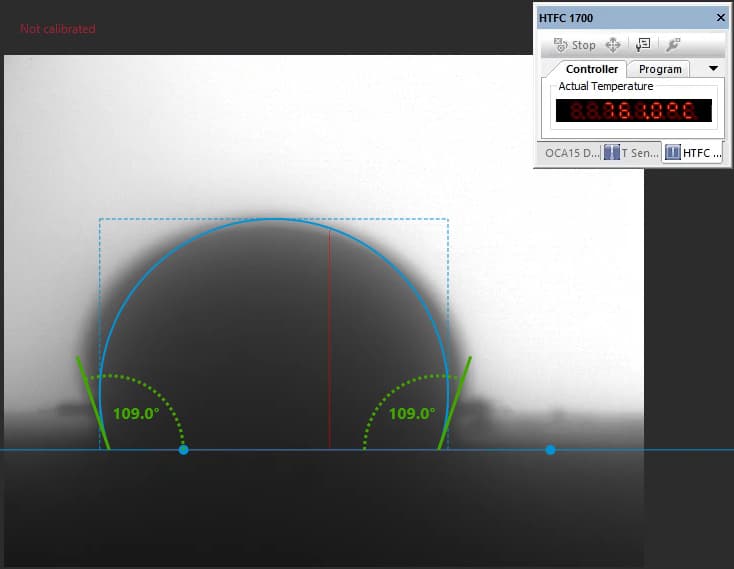
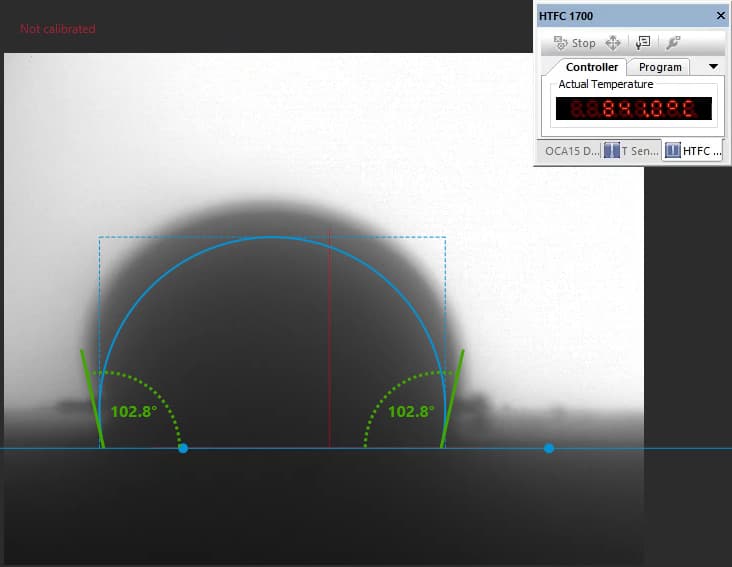
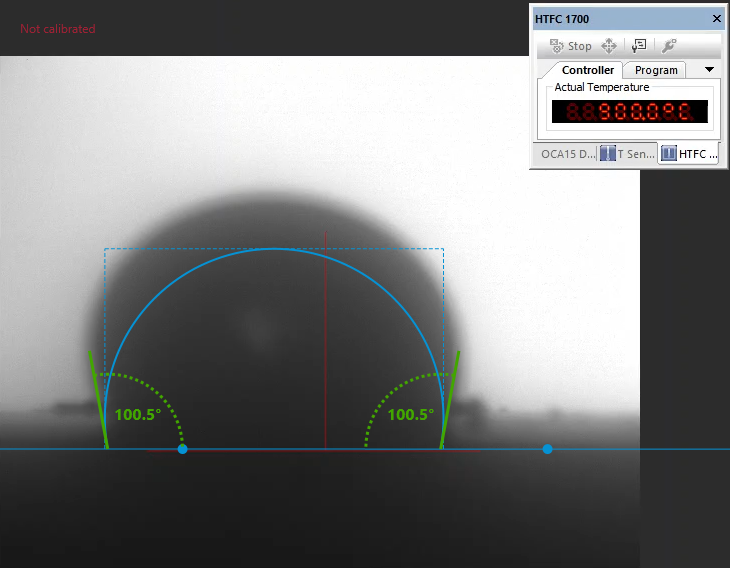
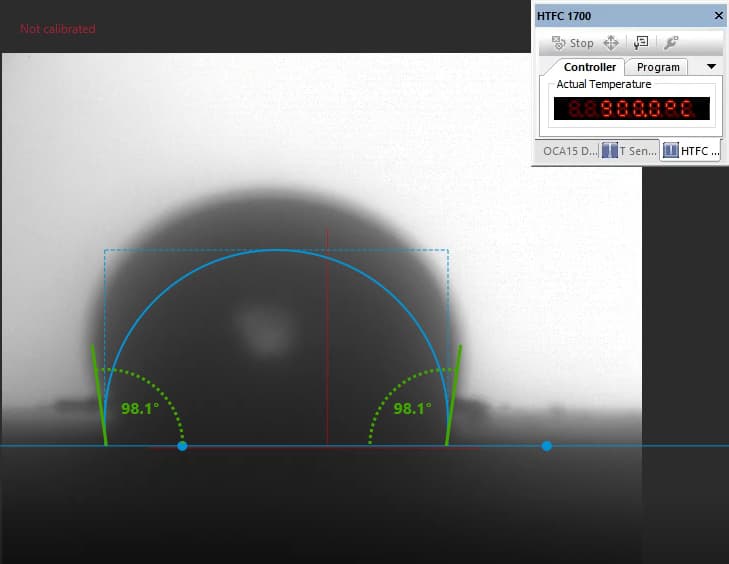
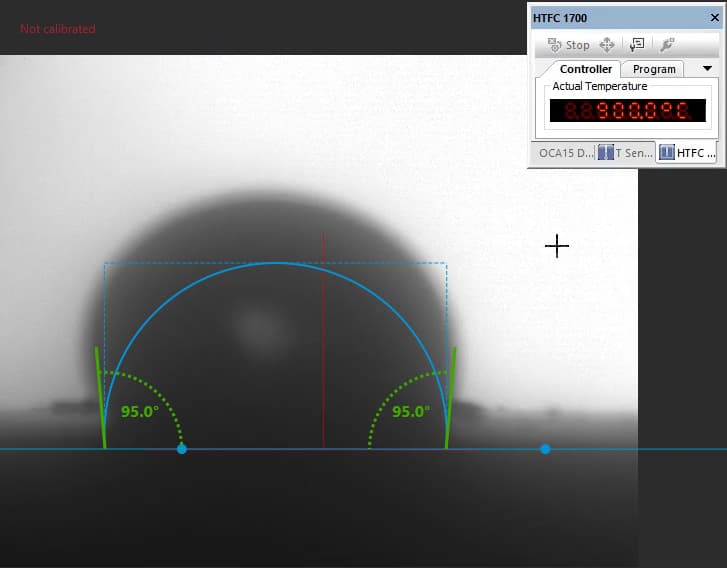
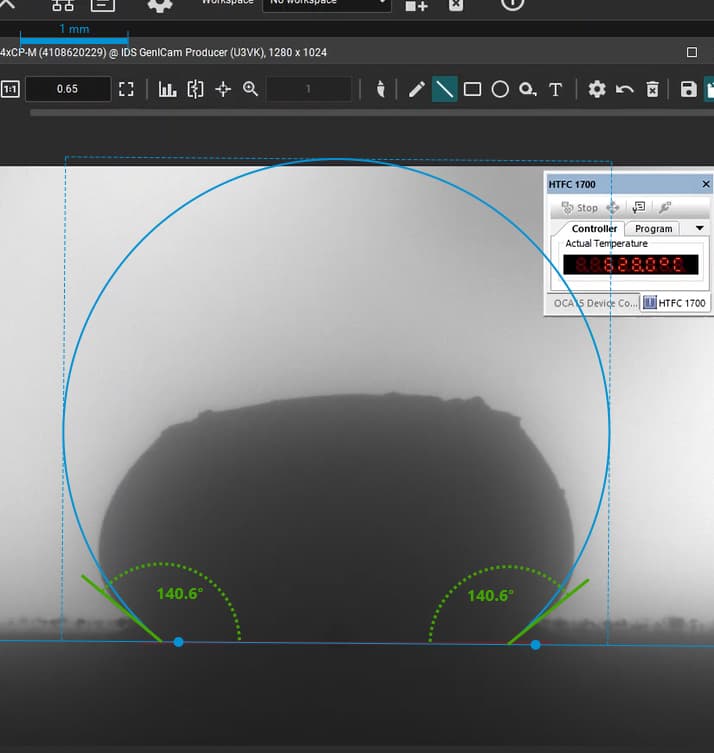
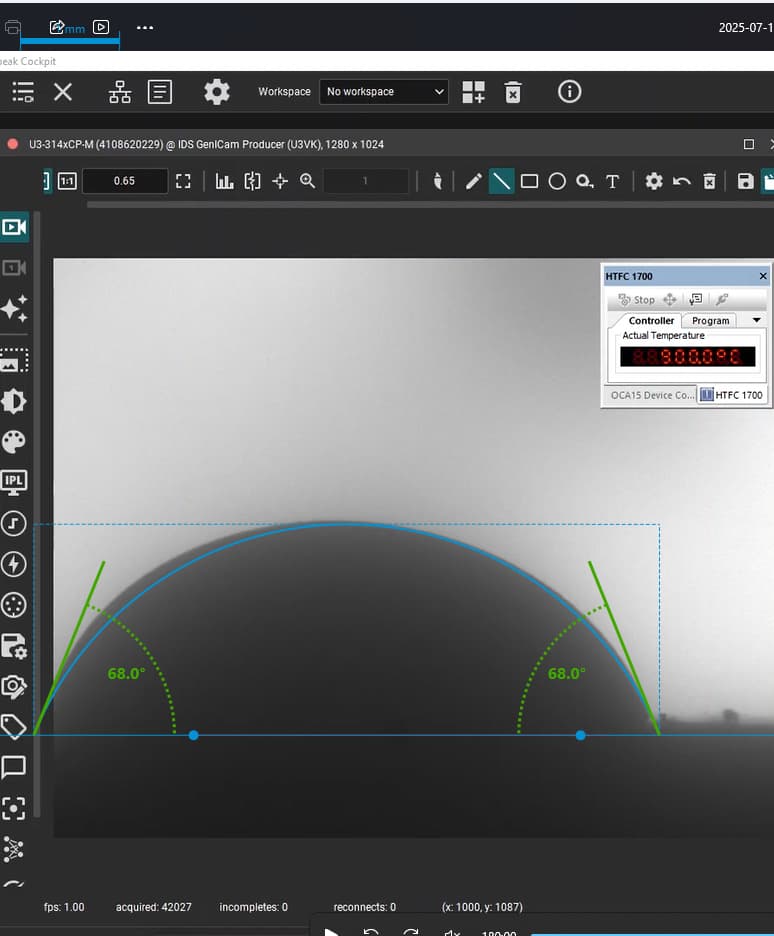
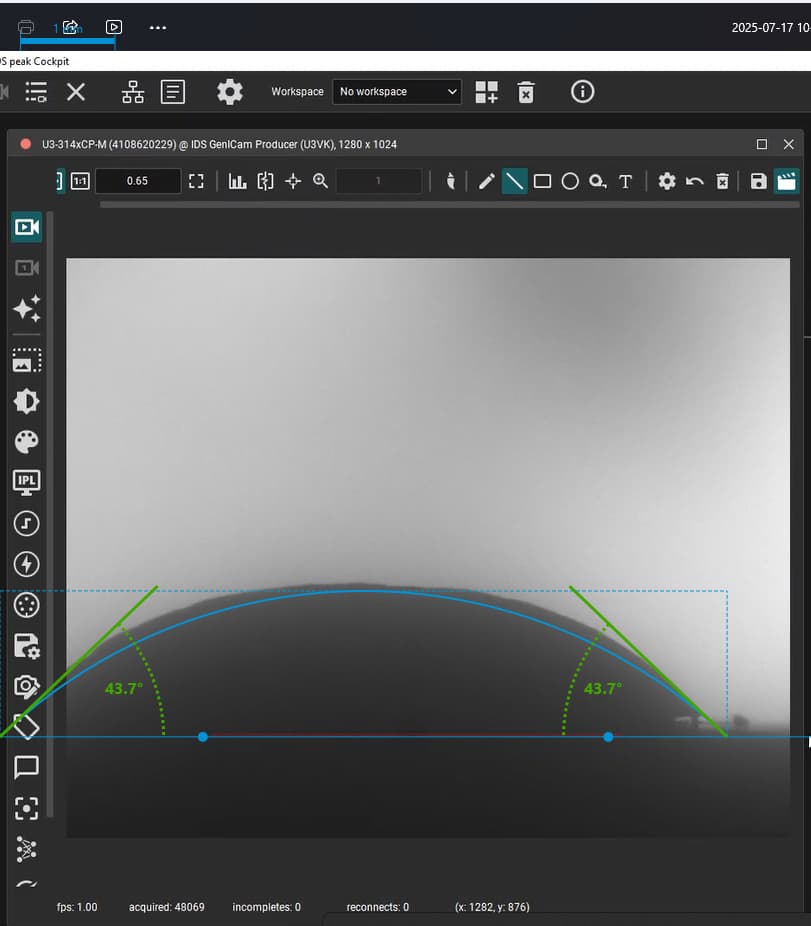
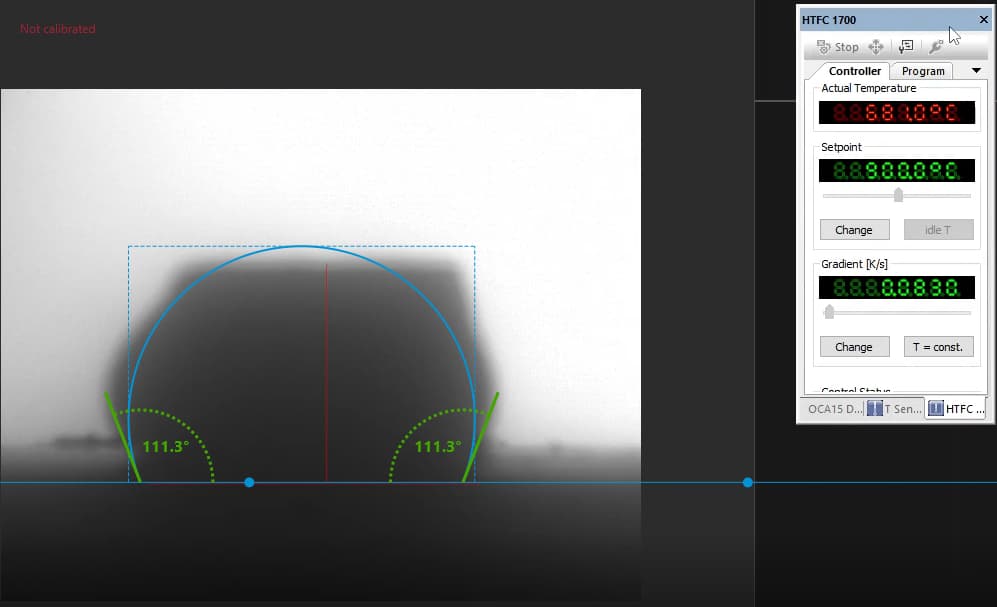
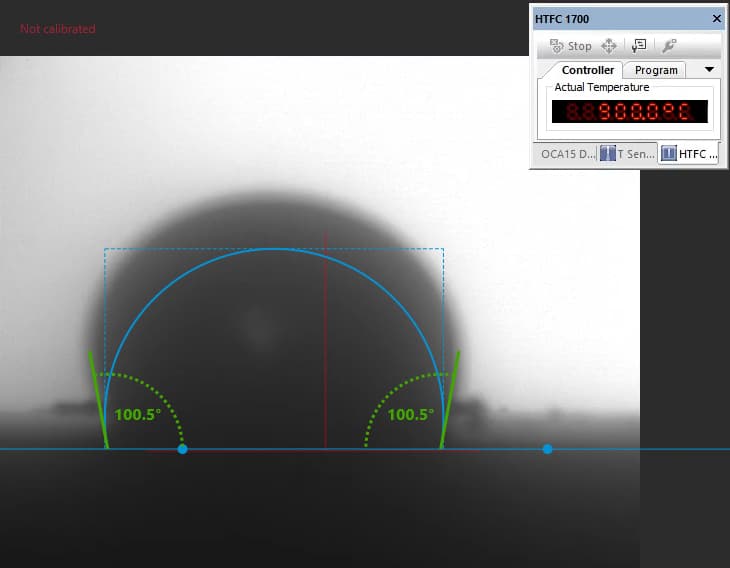
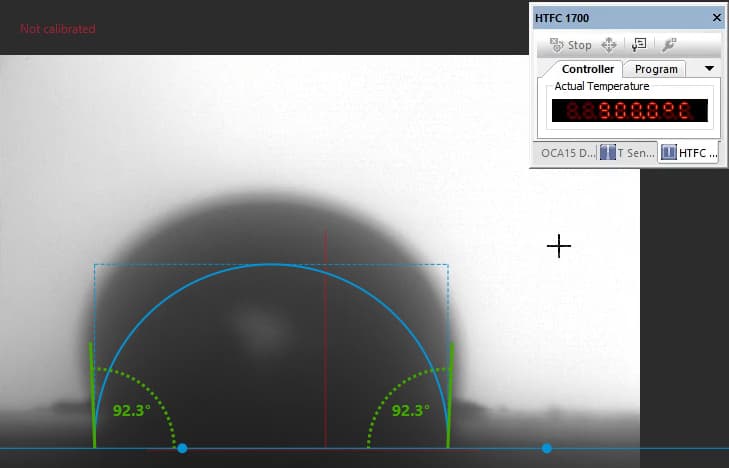
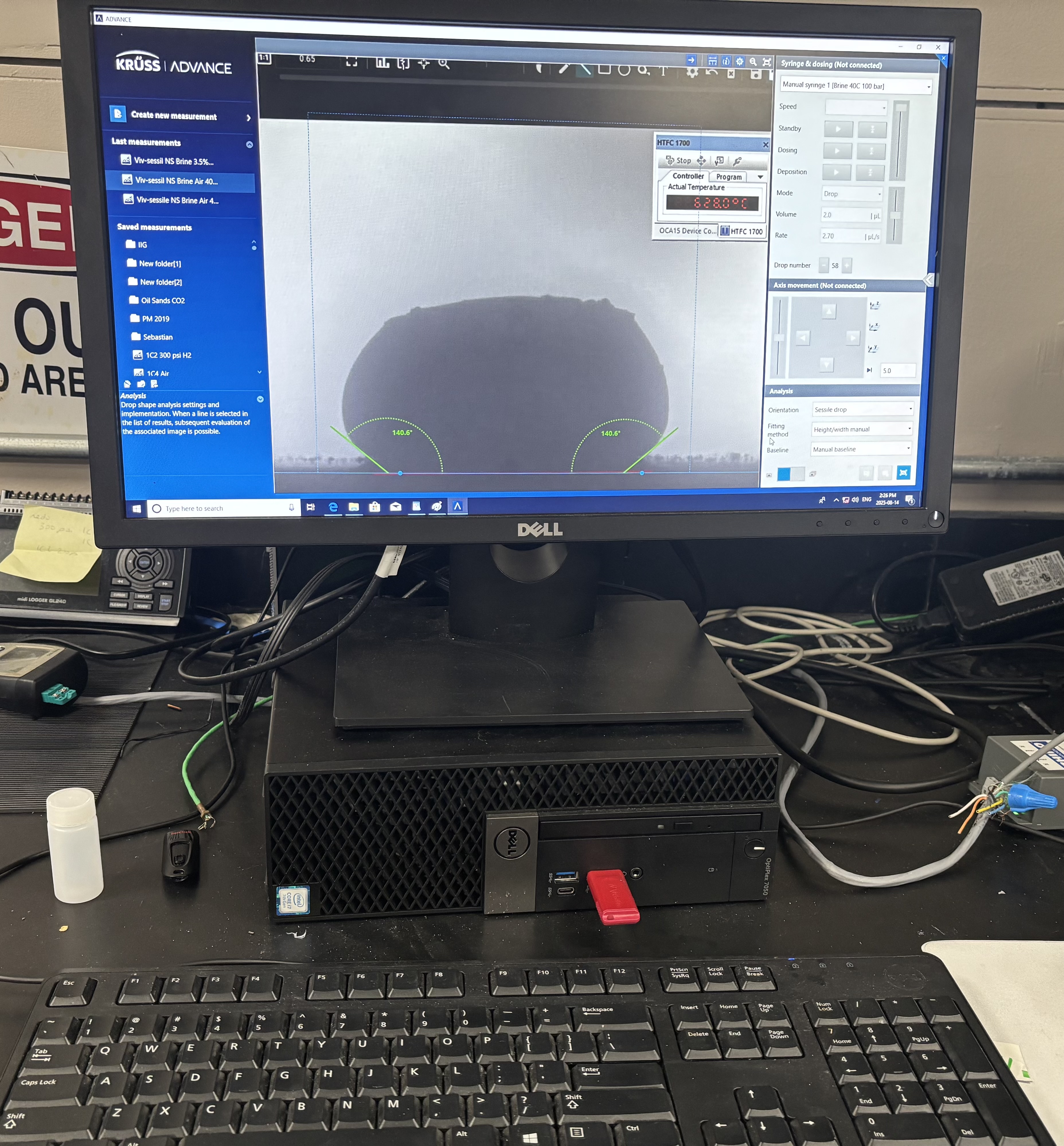
Our focus was on carbonate electrolytes, which operate optimally between 800–900 °C due to their high ionic conductivity and stability under reducing conditions. In each test, a carbon plate was placed at the base of the furnace to act as the electrode surface. On top of this, a compressed pellet of powdered electrolyte was prepared using a hydraulic press. The system was then gradually heated to 900 °C, at which point the pellet melted and formed a droplet. The degree to which this droplet spread across the carbon surface provided direct insight into the material’s wettability and, by extension, its effectiveness in enabling ionic transport within a fuel cell.
To more accurately simulate real operating conditions, the tensiometer was also equipped to introduce controlled gas flows into the furnace chamber. We conducted parallel trials in both ambient air and CO₂-rich atmospheres, the latter representing the environment of a working DCFC anode where carbon oxidation produces large quantities of CO₂. This allowed us to assess whether gas composition significantly alters electrolyte spreading and electrode–electrolyte contact behavior.
The system included a high-resolution, black-and-white scientific camera aimed directly at the reaction interface. Each experiment was filmed continuously for 2–3 hours across the heating cycle. Once complete, I processed the video data using custom Python scripts to trim heat-up phases and extract images at 10-minute intervals beginning from the melting point. These snapshots were then analyzed with specialized contact angle software available at CanmetENERGY to measure the droplet progression over time. By quantifying contact angle changes, we developed a comparative dataset of how different carbonate electrolytes performed across temperature ranges and gas environments.
View inside the tensiometer chamber where
droplets formed and spread on carbon substrates.
High-temperature tensiometer system
for electrolyte spreading studies.
From Droplets to Data
Each run generated hours of video. I developed Python scripts to process results:
video_speed.py
screen_crop.py → cropped and focused on droplet regions
video_crop.py → trimmed long videos to key reaction windows
Experiments & Findings
To evaluate candidate electrolytes for direct carbon fuel cells (DCFCs), I conducted a series of wettability tests on carbon substrates. The experiments were designed to capture how different carbonate-based electrolytes interacted with the carbon surface under both ambient and CO₂-rich conditions. Droplet images were recorded and analyzed to measure changes in contact angle over time, providing a clear metric of wetting performance.
Since this work was conducted under an NDA, I will refer to electrolytes generically (e.g., “Electrolyte 1” and “Electrolyte 2”) rather than by their exact formulations. Representative images and graphs are included to illustrate typical outcomes.
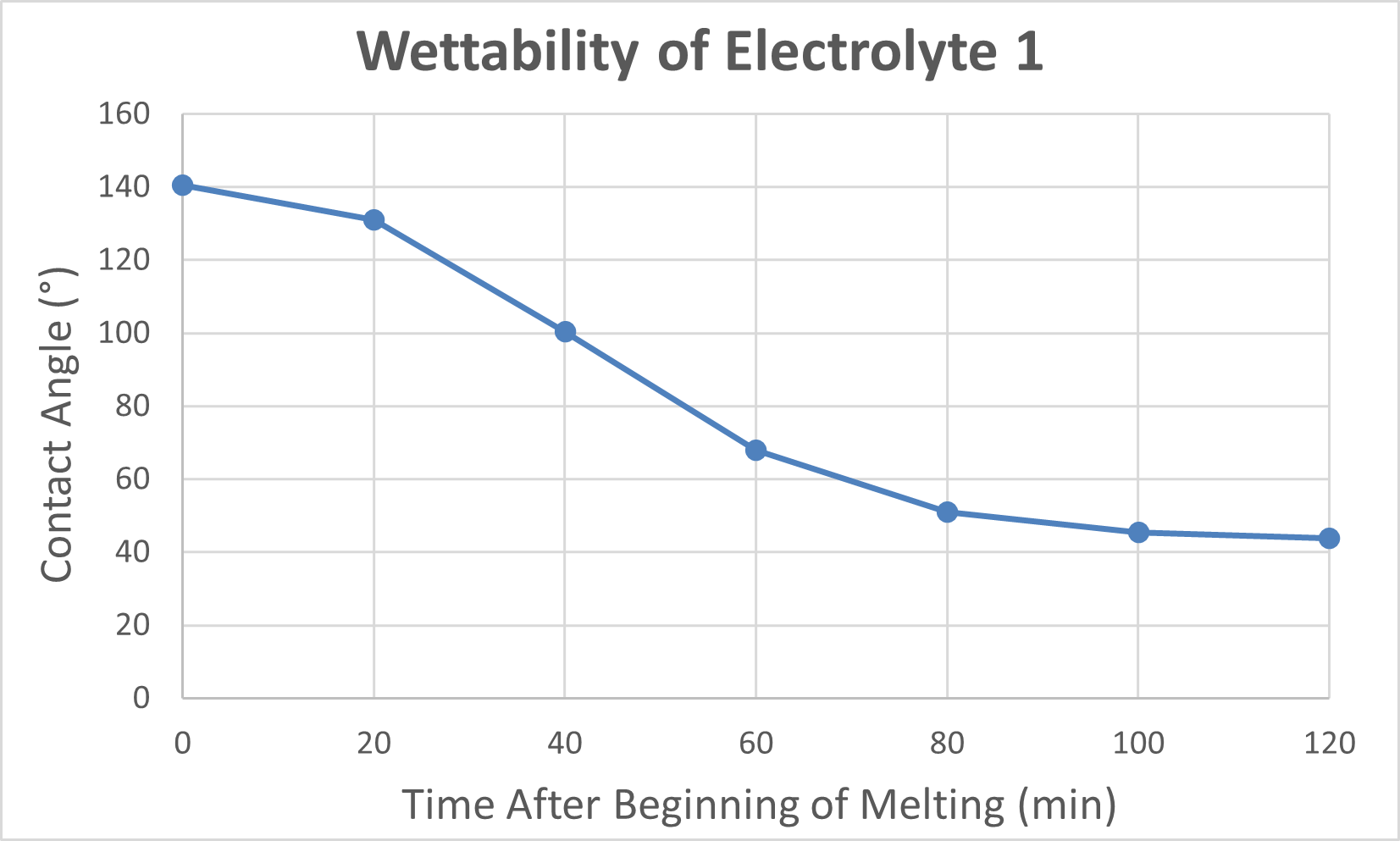
Electrolyte 1 – Strong Wetting Behavior:
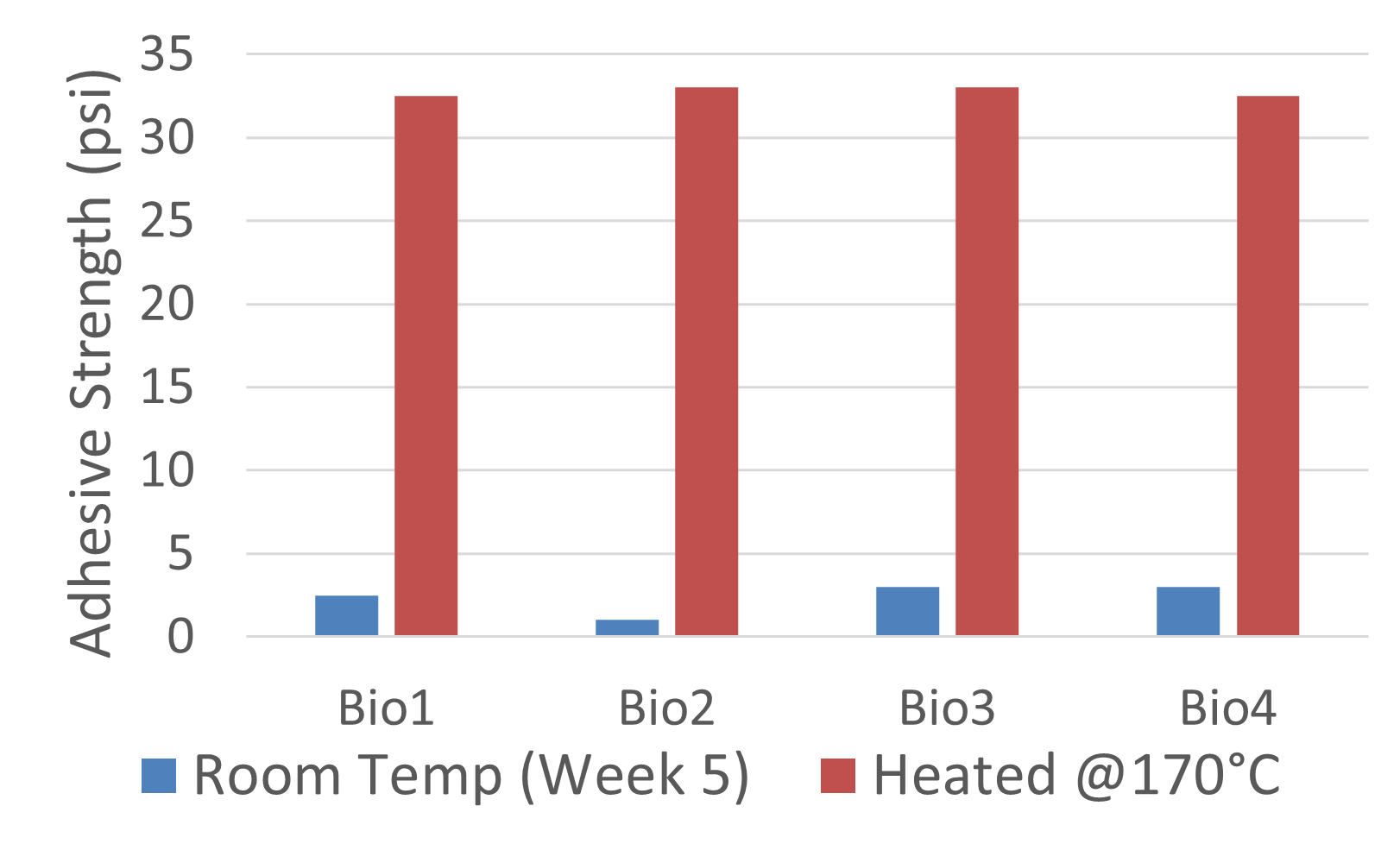
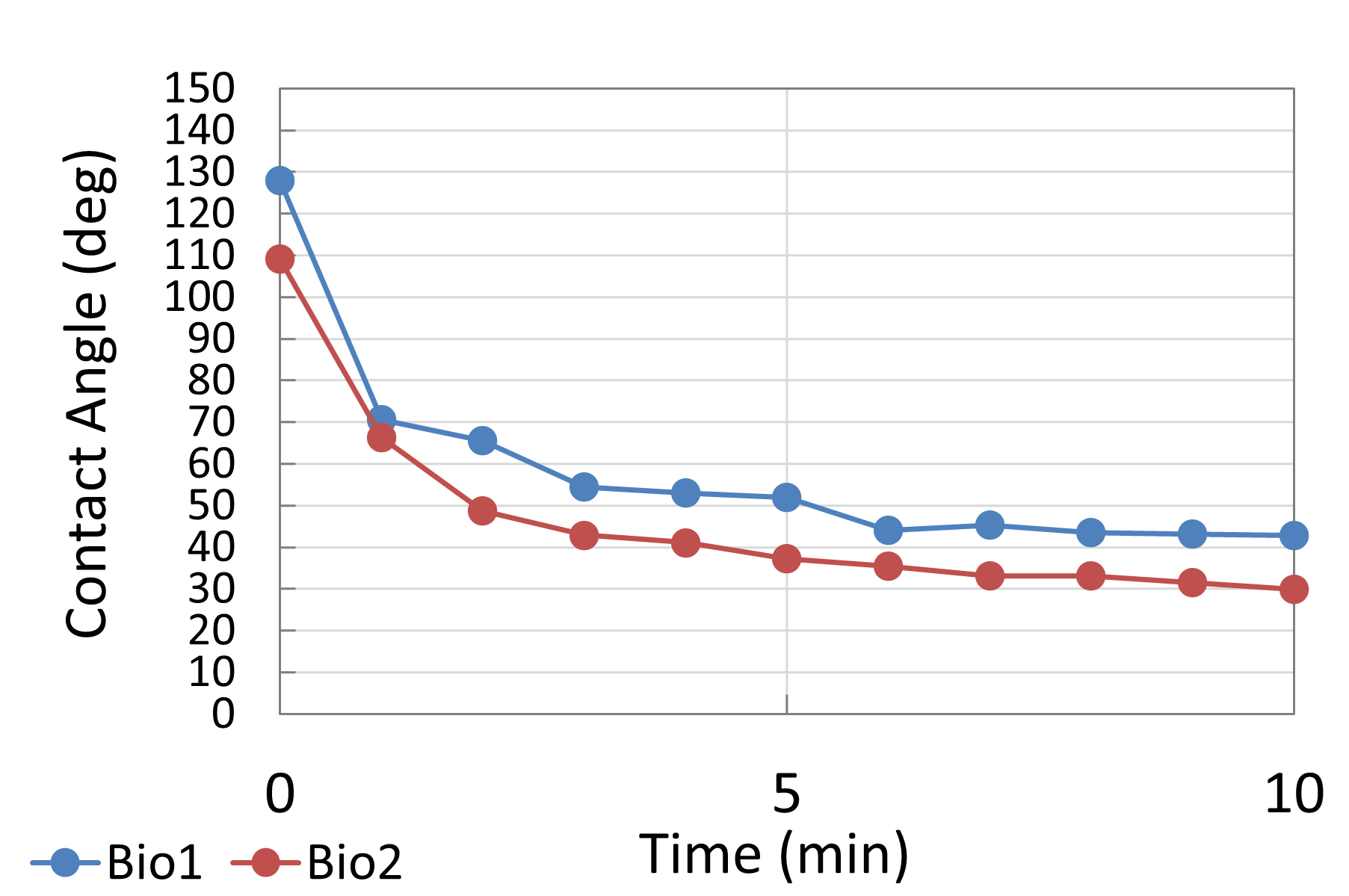
Electrolyte 1 consistently displayed favorable spreading behavior, achieving lower contact angles and maintaining stable wettability over time. This suggested strong surface interaction and effective ionic conduction pathways. I included photographs of “good wetting” droplets from this series, supported by contact angle analysis graphs that highlight the sharp decline in angle during the first few minutes of testing.
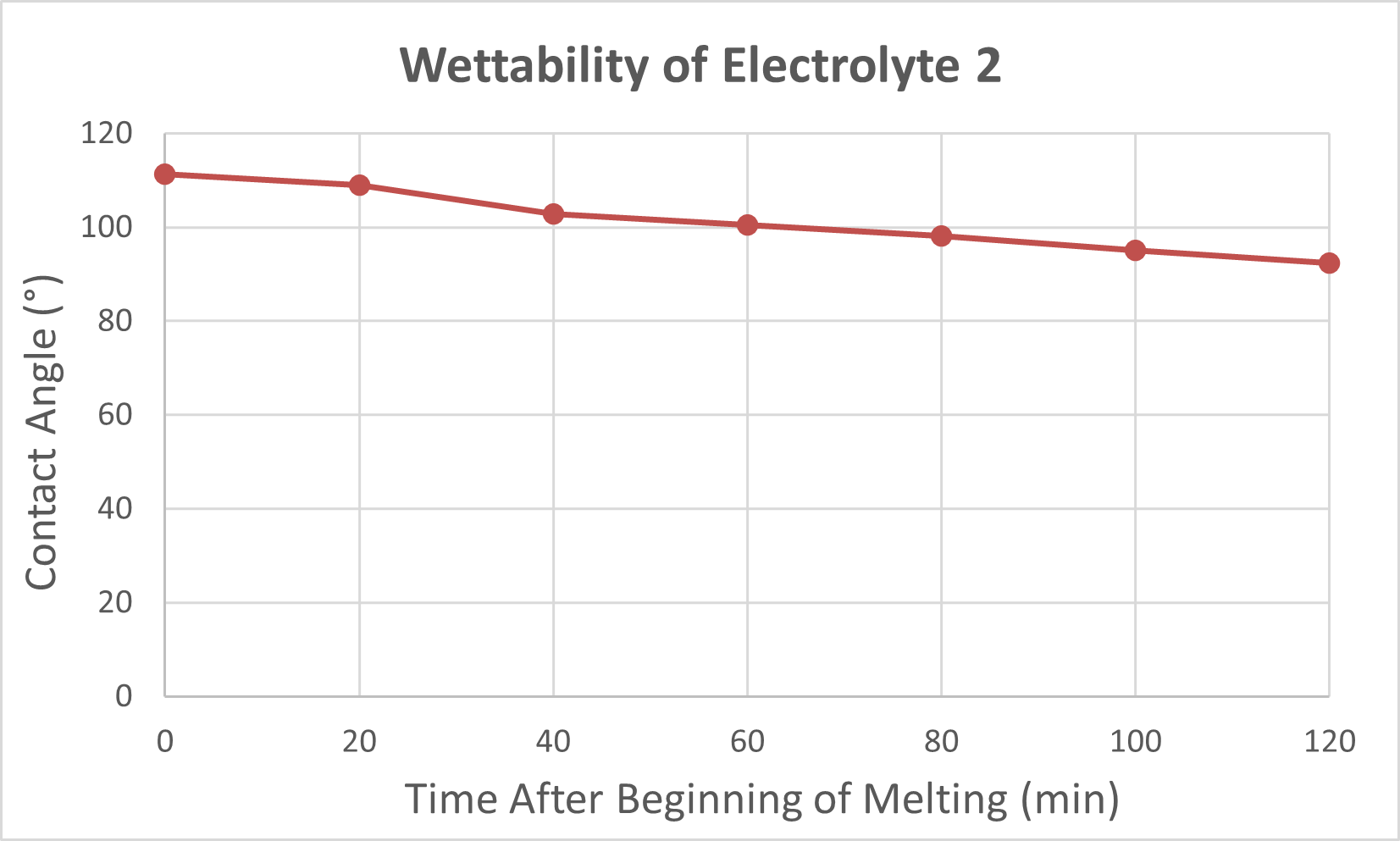
Electrolyte 2 – Poor Wetting Behavior:
In contrast, Electrolyte 2 showed poor affinity for the carbon substrate. Droplets retained high contact angles with minimal reduction over time, reflecting limited interaction with the surface. Images of these droplets illustrate “bad wetting” behavior, and accompanying graphs quantify the consistently high angles measured during testing.
Effect of CO₂ Environment:
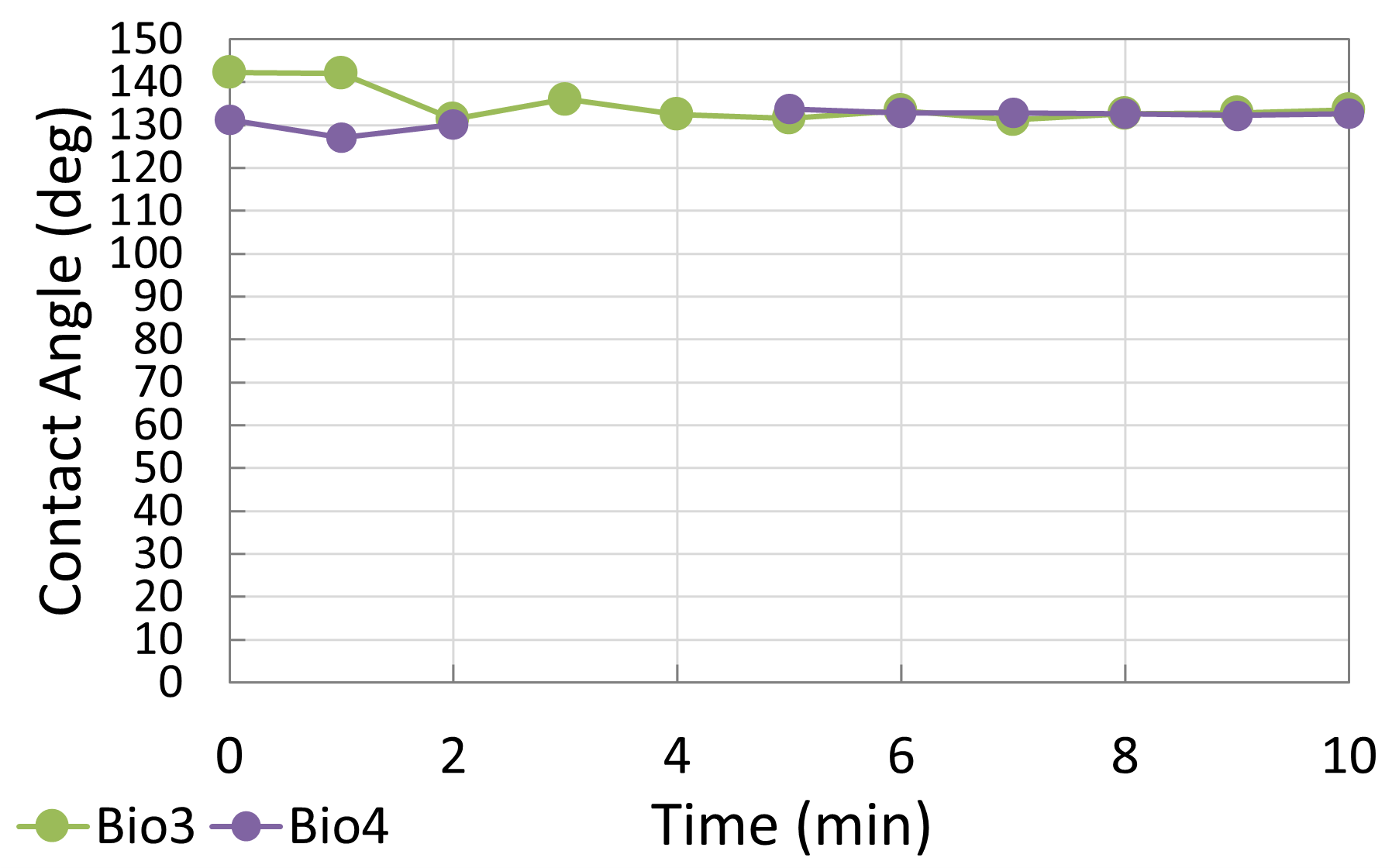
After establishing baseline behavior, Electrolyte 1 was retested under a CO₂ atmosphere to approximate operating conditions within a DCFC. The results revealed a modest increase in contact angle compared to tests in air, indicating slightly reduced wettability. I attributed this effect to surface carbonation on the biocarbon substrate, which likely altered its surface energy. While the electrolyte maintained generally good wetting, the shift emphasized the importance of considering realistic fuel cell conditions when evaluating candidate electrolytes.
Visual Data Representation:
To complement the descriptive results, I generated side-by-side Excel graphs comparing the contact angle trends of Electrolyte 1 and Electrolyte 2. These plots, together with droplet images, provide clear visual evidence of the performance differences between well-wetting and poorly wetting candidates. Overall, these findings demonstrated that while certain carbonate electrolytes can achieve strong initial wettability, their stability under a CO₂ environment must be carefully assessed to ensure long-term fuel cell performance.
Fixing Flaws, Finding Solutions
During testing, two key challenges emerged that required adjustments to ensure reliable results.First, the biocarbon surfaces occasionally displayed variations in roughness, which influenced droplet spreading behavior. To minimize this variability, I standardized sample preparation by ensuring flatter, more uniform surfaces before running trials.Second, some of the raw droplet images were difficult to analyze due to blurred edges. This made contact angle extraction less reliable. I solved this by preprocessing images through cropping and zoom adjustments, which improved edge detection and produced more consistent measurements across different runs. These refinements improved the repeatability of the experiments and ensured that the performance differences between electrolytes were due to material behavior rather than experimental artifacts.
Progression of Electrolyte 2 after video processing
Engineering Insights & Impact
The wettability experiments demonstrated that electrolyte selection plays a decisive role in enabling efficient carbon–electrolyte contact within DCFCs. Electrolyte 1, which achieved consistently low contact angles, highlighted how favorable surface interaction directly supports ionic transport and reaction kinetics. In contrast, Electrolyte 2 underscored the risks of poor spreading, where high contact angles translate into limited effective surface area and reduced electrochemical performance.
Testing in a CO₂ environment revealed an additional layer of complexity. Even the best-performing electrolyte experienced a modest reduction in wettability, likely due to surface carbonation on the carbon electrode. While the effect was not catastrophic, it reinforced the importance of validating materials under realistic fuel cell conditions rather than relying on ambient air measurements alone. These results suggest that successful DCFC operation depends not only on selecting an electrolyte with strong inherent spreading behavior, but also on understanding how its performance evolves under operating gases.
From an engineering standpoint, these findings illustrate a broader principle relevant to both fuel cells and batteries: interface behavior often determines system performance. Even small shifts in contact angle can cascade into changes in ionic conductivity, polarization losses, and overall efficiency. For recruiters and cell engineers, this project demonstrates my ability to design controlled experiments, identify subtle surface–electrolyte interactions, and extract engineering insights that inform material selection and system optimization.


In conclusion, my work contributed to CanmetENERGY’s broader DCFC development efforts by narrowing down promising electrolyte–carbon pairings while highlighting the critical need for CO₂-environment validation. More broadly, it strengthened my understanding of electrolyte behavior at extreme conditions and sharpened my experimental and analytical skills in electrochemical systems. These capabilities align directly with my long-term goal of contributing to next-generation cell and battery technologies, where interface design and electrolyte optimization are central challenges for advancing clean energy solutions.
“good wetting” droplet showing flat profile and strong surface interaction.
Progression of Electrolyte 2 after video processing
Want to work with an engineer who builds, tests, and iterates from first principles?
Designed to showcase not just what I can build — but how I think, test, and iterate like an engineer in the field.
Software used to measure droplet contact angles frame-by-frame.
Sped-up footage of Electrolyte 1 melting and
spreading across the carbon substrate.
“poor wetting” droplet retaining high curvature and weak surface contact.
Sped-up footage of Electrolyte 1 melting and
spreading across the carbon substrate.
Sped-up footage of Electrolyte 2 retaining poor
wetting behavior across the test cycle.
.JPEG)

.jpg)







![[digital project] image of factory automation](https://cdn.prod.website-files.com/686ac8c2cf315121feffaa5e/6881728d32e8bab0d34175fa_5.png)
.jpg)
.JPEG)
.png)

.png)
.JPEG)



.gif)


%20(2).jpg)